Fruit fly model provides inside look into progression of rare human disease
Elena Gracheva in Chao Zhou’s lab uses optical coherence microscopy to monitor disease progression in fruit fly cardiovascular system
Retinal vasculopathy with cerebral leukoencephalopathy and systemic manifestations (RVCL-S) is a rare disease that causes deterioration of small vessels, and therefore, affects the eyes, brain, liver and other organs with many small vessels. Symptoms of the disease usually appear in individuals between ages 35-55, but since there is no cure, patients with RVCL-S have a shorter life expectancy.
Researchers in the McKelvey School of Engineering at Washington University in St. Louis led a multi-institutional team in developing a fruit-fly-based model of RVCL-S that could be used as a screening platform to test new drugs, to search for drug targets and to facilitate future research. Results of the research were published March 6, 2026, in eLife.
Elena Gracheva, a staff research scientist in the lab of Chao Zhou, professor of biomedical engineering, said that fruit flies’ genome contains genes similar to 75% of human disease-causing genes. Fruit flies represent an efficient system to study age‑related changes across the lifespan, the researchers said.
“Traditionally, researchers have used mouse models or cell cultures for this research, but they either do not reliably capture the nature of a human disease or are not efficient for primary screening purposes,” Zhou said. “We can potentially have thousands of flies to do large quantity primary screening. This may be a very important platform to help find a cure for RVCL.”
Research into RVCL-S has a long history at WashU. In research published in Nature Genetics in 2007, John Atkinson, MD, the Samuel Grant Professor of Medicine and professor of molecular microbiology at WashU Medicine, found that RVCL-S is linked with variants of the TREX1 gene. A shortened version of TREX1 protein breaks down DNA when it penetrates a cell’s nucleus. Following Atkinson was Jonathan J. Miner, MD, PhD, previously at WashU Medicine and now an associate professor of medicine at the RVCL Research Center at the University of Pennsylvania and a co-author of the paper in eLife.
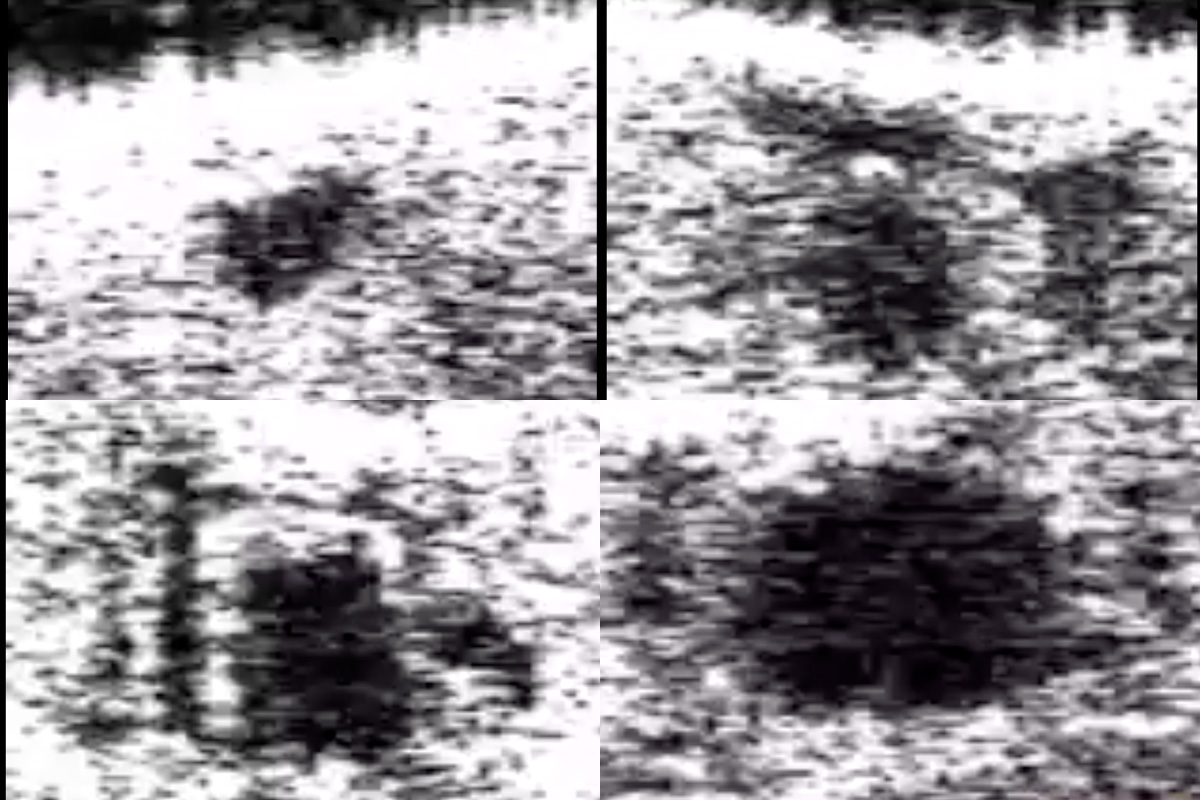
In the new research in Zhou’s lab, Gracheva and a team of students identified the gene in fruit flies, cg3165, by similarity in the gene sequence to the human TREX1 gene and predicted it to have a similar function. Since small blood vessels are affected in patients with RVCL-S, they looked to recreate the disease in fruit flies. They created three models of fruit flies: one with cg3165 removed; one with a normal human TREX1 gene added; and one with a mutated TREX1 gene. They used optical coherence microscopy (OCM), a powerful imaging method in Zhou’s lab, to monitor changes in the fruit flies’ cardiovascular system, a single tube — or heart tube — running nearly the length of their bodies.
“When we deleted the cg3165 gene in the fruit fly, the diameter of the heart tube became much smaller, so it was affected significantly, similar to the vascular system in humans with this disease,” Gracheva said. “In the other two models with the normal gene and the mutated gene, the heart tubes had a larger diameter.”
Gracheva said as the flies age, their heart tube and their movement deteriorate, particularly in the flies carrying the mutated gene. Removing the gene also led to impaired movement and a shorter lifespan.
RVCL affects various organs in the body, including the brain. Survival of fruit flies depends on their motor functions, which is coordinated by the central nervous system processing external signals and sending instruction and muscles performing certain actions. To study this, the team put the different models of fruit flies in small containers to observe their climbing behavior.
“Typically, for normal flight, fruit flies will fly up to the top of a container,” Zhou said. “But with the mutated gene, the flies were not as active as the normal ones, and they stayed on the bottom of the container, suggesting that the TREX1 gene affects neurological networks and muscle tissue condition in the flies.”
This robust phenotypic system for RVCL-S could help other researchers who are looking for a cure or to repurpose another drug for the disease, the researchers said.
“We’re not looking to replace the mouse model,” Gracheva said. “The flies would allow us to filter thousands of molecules of chemical compounds and narrow the number of candidates.”
Gracheva E, Matt A, Wang F, Hsin R, Liang H, Ouyang X, Ding J, Miner JJ, Zhou C. Drosphila melanogaster model of RVCL-S demonstrates age dependent disease progression. eLife, reviewed preprint, March 6, 2026. DOI: https://doi.org/10.7554/eLife.109595.1
This research was supported with funding from the National Institutes of Health (R01-HL156265 (CZ), R01AI143982, R01NS131480 (JJM); Clayco Foundation Innovative Research Award (CZ); gift from the Clayco Foundation (JJM); National Science Foundation Graduate Research Fellowship Program (AM); Washington University in St. Louis startup fund (CZ); Penn Colton Center for Autoimmunity pilot award (JJM); Penn RVCL Sisters Fund (JJM).





