New research on cellular redox reactions sheds light on the path to neurodegenerative diseases
Yifan Dai and colleagues are reshaping our understanding of disease origins and revealing how these condensates can tip cells from normal behavior to dysfunction
The mechanics of the onset of cancer or neurodegenerative disorders like Alzheimer’s disease or ALS remain a mystery. Scientists associate these diseases with an increase in unstable molecules called reactive oxygen species (ROS) in cells, but don’t fully know why they form or why these molecules might pose a problem. They are also beginning to determine which parts of cells are to blame to produce ROS.
Studies have shown that one possible source of these free oxygen radicals is mitochondria, membrane-bound structures in the cell. And previous work done by Yifan Dai, assistant professor of biomedical engineering in the McKelvey School of Engineering at Washington University in St. Louis, found yet another origin for ROS: small agglomerations of mostly proteins called biomolecular condensates.
In a new paper published in the Journal of the American Chemical Society (JACS), Dai and Anthony A. Hyman from Max Planck Institute of Molecular Cell Biology and Genetics dive deeper. They address how these biomolecular condensates create oxygen-based radicals without the presence of enzymes and uncover what are the chemical origins of the radical sources. Such findings provide the molecular basis for the study of the roles of condensates in cellular physiology and diseases.
The electrochemistry in condensates
Mitochondria, long considered the primary source of free oxygen radicals, are bound by membranes and need enzymes to catalyze the formation of ROS.
In 2023, Dai and team found that biomolecular condensates don’t need to play by the same rules. They are not bound by membranes, nor do they need enzymes to kickstart redox reactions. Instead, free oxygen radical-forming reactions in condensates are spontaneous.
What drives such spontaneity? The exact sources and the mechanisms for the processes were a mystery. The answer lies in the nature of the condensate itself. Unequal distribution of charged ions within the cluster creates a discrete interfacial electric field between the condensate and the rest of the cell. Such marked differences in charges from positive and negative ions result in electrical fields, which then trigger oxidation reactions.
The condensates also have an alkaline environment, which helps compounds lose hydrogen molecules, a process known as deprotonation. Ready candidates for deprotonation are already nearby: the proteins in the condensate. Proteins contain amino acids, one of which, tyrosine, is especially vulnerable to losing its hydrogen molecule and in the process, releasing a free electron.
From an alkaline environment to problems
As it turns out, the path to rogue behavior from the ROS or free oxygen radicals is long yet systematically facilitated by biomolecular condensates.
First, the electrochemical environment of the condensate, with its elevated pH, facilitates tyrosine oxidation. The free electron that results from the process can combine with oxygen to form a superoxide radical and then generate hydrogen peroxide. The other pathway to hydrogen peroxide is through the combination of two OH radicals released by direct water oxidation (see figure).
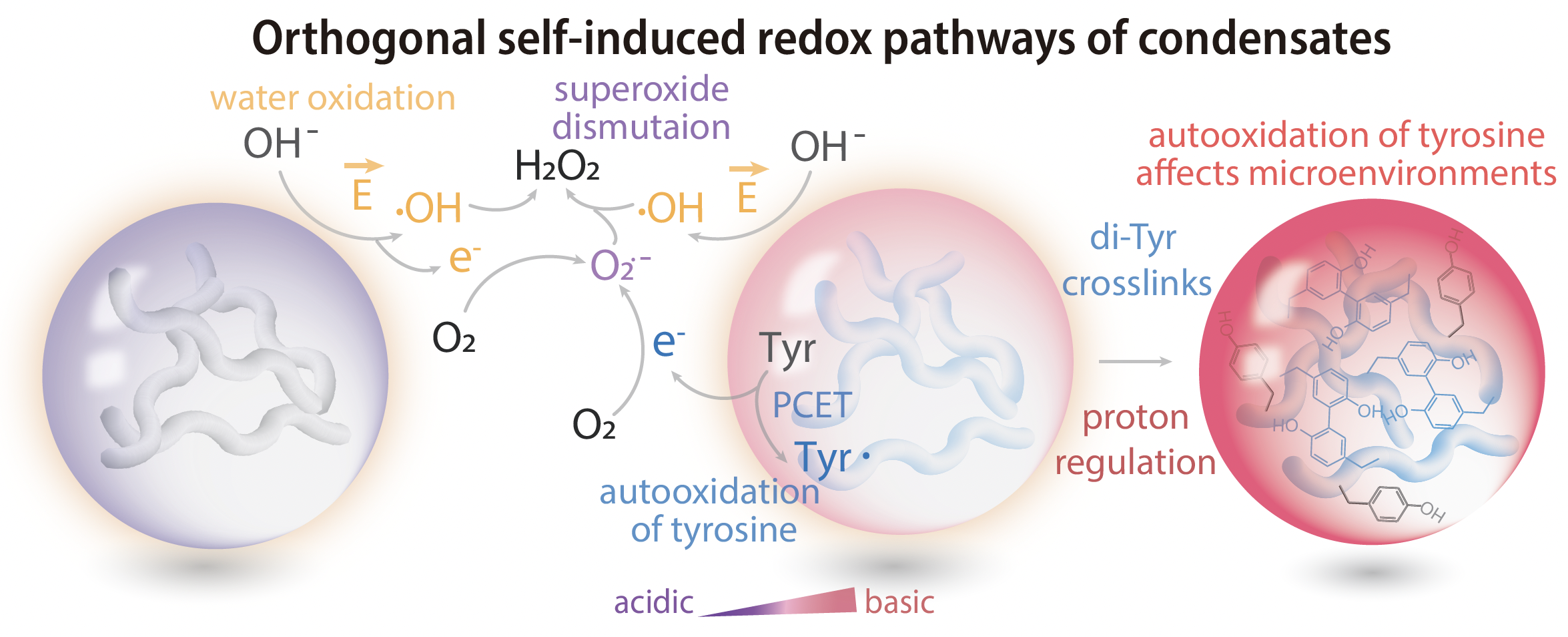
How hydrogen peroxide forms from tyrosine.
No matter which way it forms, hydrogen peroxide is a problem because it can contribute to protein damage and accelerate the pathway to neurodegenerative diseases.
Another important finding from Dai and his team is that the condensate environment is not static, but ages with time. Since hydrogen peroxide formation consumes protons, the condensate steadily becomes even more alkaline. In essence, the condensate both triggers chemical reactions and changes its own internal environment in the process. It means condensates are dynamic living forms and not simply passive aggregates of protein molecules.
Application to nitrogen cycling
In another recent paper in JACS, Dai and Richard N. Zare from Stanford University explained how biomolecular condensates, with an ability to trigger spontaneous oxidation processes, can help the nitrogen cycle through its different chemical forms. Nitrogen in its various forms is necessary for a variety of cell functions. Nitric oxide, for example, helps regulate blood pressure.
Just as in the tyrosine oxidation example, the paper showed how nitrogen cycling can proceed without the help of enzymes. In such a case, condensates modulate reactive nitrogen species.
Both papers illustrate the importance of an enzyme alternative: an electrical field created by an imbalance of ionic charges at the interface between the condensate and the cell.
“An electrical field, nanoscopic or mesoscopic, is the driving force for any kind of chemistry you do whether that’s in a battery or in soft living matter,” Dai said.
Exploring cell behavior from the lens of physical and electrochemistry is important, Dai said, because it can complement knowledge of cell genomic sequencing to better understand the basic triggers of cancer and many other diseases.
“We are beginning to dive deeper, to explore the many things we don’t know about cell chemistry and biology,” Dai said, “there are so many downstream chemical activities that are happening that we’re still learning about.”
The two papers point to a new direction in the fields of cell biology and chemistry: biomolecular condensates as active centers of chemical reactions. When all goes well, condensates regulate protein production and repair DNA. But when things go awry, these same cellular structures can cause diseases. Researching their behavior is an essential step toward learning when and how these biomolecular condensates misbehave. “There are many open questions in this field, and we are excited about these emerging areas.” Dai said.
Song X, Lyu L, Li C, Ma Y, Zhou Y, Dai Y, Zare RN. Biomolecular Condensates Power Nitrogen Cycling via Concurrent Redox Activities. Journal of the American Chemical Society, Vol 148/Issue 10, March 4, 2026. DOI: https://pubs.acs.org/doi/10.1021/jacs.5c22593.
This research was supported by the Alzheimer’s Association (AARG-25-1486936) and McKelvey School of Engineering and Center for Biomolecular Condensates at Washington University; the Air Force Office of Scientific Research through the Multidisciplinary University Research Initiative program (AFOSR FA9550-21-1-0170) and the Sustainability Accelerator program of the Doerr School of Sustainability at Stanford University (GHG-0030-R.N.Z.).
Yu W, Zhou Y, Yang L, Yan X, Smukowski SN, Ma Y, Fan J, Goo YA, Hyman AA, Dai Y. Origins of Intrinsic Redox Activity of Biomolecular Condensates. Journal of the American Chemical Society, March 25, 2026. DOI: 10.1021/jacs.6c01750
This research was supported by Alzheimer’s Association (AARG-25-1486936); Körber-Stiftung (Germany), the Max Planck Society (Germany) and the NOMIS Foundation (Switzerland).




