Focused ultrasound technique leads to release of neurodegenerative disorders biomarkers
Research in Hong Chen’s lab could facilitate diagnosis of Alzheimer’s disease
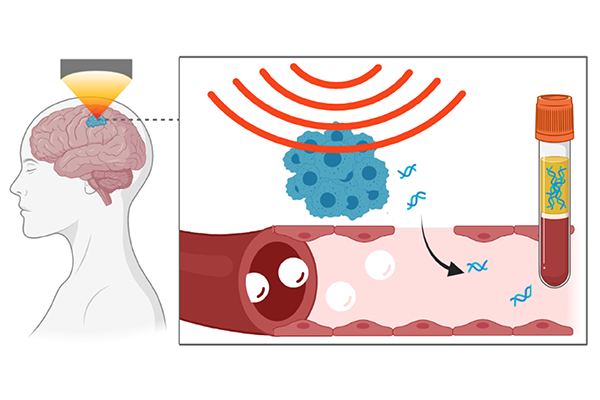
Several progressive neurodegenerative disorders, including Alzheimer’s disease, are defined by having tau proteins in the brain. Researchers are seeking to identify the mechanisms behind these tau proteins to develop treatments, however, their efforts to detect biomarkers in blood has been hampered by the protective blood-brain barrier.
At Washington University in St. Louis, new research from the lab of Hong Chen, associate professor of biomedical engineering in the McKelvey School of Engineering and of radiation oncology in the School of Medicine, and collaborators found that using focused-ultrasound-mediated liquid biopsy in a mouse model released more tau proteins and another biomarker into the blood than without the intervention. This noninvasive method could facilitate diagnosis of neurodegenerative disorders, the researchers said.
Results of the work, the first to open the door for noninvasive and targeted diagnosis and monitoring of neurodegenerative disorders with focused-ultrasound-mediated liquid biopsy, are published in Radiology Jan. 31, 2023.
The method, known as sonobiopsy, uses focused ultrasound to target a precise location in the brain. Once located, the researchers inject microbubbles into the blood that travel to the ultrasound-targeted tissue and pulsate, which safely opens the blood-brain barrier. The temporary openings allow biomarkers, such as tau proteins and neurofilament light chain protein (NfL), both indicative of neurodegenerative disorders, to pass through the blood-brain barrier and release into the blood.
Chen teamed with co-senior author Arash Nazeri, MD, assistant professor of radiology at the School of Medicine’s Mallinckrodt Institute of Radiology (MIR). They collaborated with Tammie LS Benzinger, MD, PhD, professor of radiology at MIR and professor of neurological surgery and of biology and biological sciences; Eric Leuthardt, MD, professor of neurosurgery at the School of Medicine and of biomedical engineering in McKelvey Engineering; as well as first author Christopher Pham Pacia, who earned a doctorate in biomedical engineering from Washington University earlier this year; Jinyun Yuan, a research scientist in Chen’s lab; and Yimei Yue, a research technician in Chen’s lab.
Chen, Leuthardt, Pacia, and other collaborators have been working on the sonobiopsy technique for several years, first with biomarkers for human brain cancer in preclinical models. Other liquid biopsy methods used to detect biomarkers for neurodegenerative disorders have multiple challenges, including lacking anatomical information on the location of the protein release, rapid clearance from the fluids and a filtering process by the blood-brain barrier. Chen said sonobiopsy is an emerging technique with the potential to address these and other challenges.
In the new research, the team first took blood samples from young mice with abnormal tau proteins in the brain, or tauopathy, receiving either sonobiopsy or sham treatment. They found that sonobiopsy resulted in a 1.7-fold-increase in the normalized phosphorylated pTau-181 tau protein levels and a 1.4-fold increase in normalized pTau-231 compared with the control mouse group that had not had sonobiopsy. In a follow-up study, they performed targeted sonobiopsy by targeting either the hippocampus or cerebral cortex in the early neurodegenerative stages of the tauopathy model and took blood samples before and after sonobiopsy. The targeted sonobiopsy resulted in a 2.3-fold increase in NfL protein, a secondary biomarker for neurodegenerative diseases in the treated mice compared with the control group.
“In our proof-of-concept study, we sought to determine whether sonobiopsy is able to release phosphorylated tau species and NfL into the bloodstream by opening the blood-brain barrier,” Chen said. “This demonstration showed that sonobiopsy significantly enhanced the release of pTau proteins and a secondary marker of neurodegeneration into the bloodstream for noninvasive diagnosis for neurodegenerative diseases.”
Nazeri said tauopathies such as Alzheimer’s disease are similar to brain tumors.
“While brain tumor behavior and treatment response are dictated by the specific mutations they harbor, the tau protein shows great heterogeneity in the pattern of phosphorylation as well as other post-translational modifications,” Nazeri said. “Current PET imaging and recently developed plasma biomarkers are sensitive to detect tauopathies even in early stages. Sonobiopsy could potentially play a role to further characterize the specific strains of tau protein present in the brain for personalized treatment of people with Alzheimer’s disease and other tauopathies.”
Going forward, the team will examine the qualitative effects of sonobiopsy on plasma biomarkers and characterize the effects of focused ultrasound parameters and determine an optimal blood collection time, as well as determining how sonobiopsy can be applied to release larger brain-derived protein biomarkers.
Pacia C, Yuan J, Yue Y, Leuthardt EC, Benzinger TLS, Nazeri A, Chen H. Focused ultrasound mediated liquid biopsy in a tauopathy mouse model. Radiology. Jan. 31, 2023. https://doi.org.10.1148/radiol.220869
This research was supported with funding from the Radiological Society of North America Research & Education Foundation/Canon Medical Systems USA Inc. (RR1953); and the National Institutes of Health (R01EB027223, R01EB030102, R01MH116981, T32NS115672).




